AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Argon molar mass12/31/2023 New York: Springer-Verlag New York, Inc., 2003.Ĭonnelly, Neil G., Ture Damhus, Richard M. "Atomic Screening Constants from SCF Functions." Journal of Chemical Physics, volume 38, number 11, 1963, pp. 2686–2689. 
"Explicit Periodic Trend of van der Waals Radii." The Journal of Physical Chemistry, volume 96, number 23, 1992, pp. 9194–9197. Berlin: Springer-Verlag, 1978.Ĭhauvin, Remi. Photoemission in Solids I: General Principles. Calculation and Conversion to Pauling Units." Journal of ChemicalĮducation, volume 65, number 1, 1988, pp. 34–41. "Revised Mulliken Electronegativities: I. 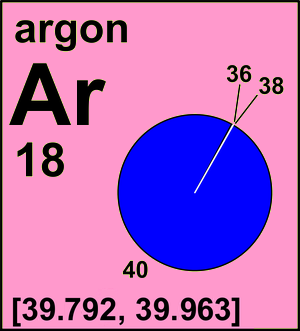
"Van der Waals Volumes and Radii." The Journal Secondary Reference Points." Metrologia, volume 33, number 2, 1996, pp. 133–154. "Recommended Values of Temperature on the International Temperature Scale of 1990 for a Selected Set of "Elemental Etymology: What's in a Name?" Journal of Chemical Education, volume 62, number 9, 1985, pp. 787–788. "Binding Energies in Atomic Negative Ions: III." Journal of Physical and Chemical Reference Data, volume 28, number 6, 1999, pp. 1511–1533.īall, David W. Meteoritic and Solar." Geochimica et Cosmochimica Acta, volume 53, number 1, 1989, pp. 197–214. "The Definition of Electronegativity and the Chemistry of the Noble Gases." Journal of "Electronegativity Is the Average One-Electron Energy of the Valence-Shell Electrons in Ground-State Free Atoms." Journal of New York: Oxford University Press, 1992.Īllen, Leland C. The volume of air is 500 mL at 22☌.Next to a value above to see complete citation information for that entry)Īlbright, Thomas A., and Jeremy K. A sample of air is trapped in a cylinder that is fitted with a moveable piston. 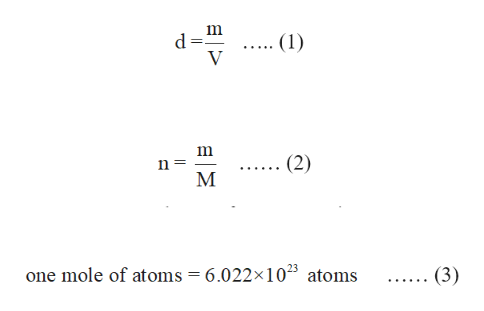
What pressure will it now exert? (Recall that in the combined gas law, pressure and volume can be in any units, so long as they are the same units for both pressures and the same units for both volumes.) 5. The gas is cooled and compressed to a volume of 50. ☌, 0.85 atm? A sample of argon gas occupies 100. g of Ar occupy at 0 ☌, 760, mm Hg? (Hint: what's special about 0 ☌, 760. (a) What volume will be occupied by this same 10. pressure and volume can be in any units, so long as they are the same units for both pressures and the same units for both volumes.) 5. What pressure will it now exert? (Recall that in the combined gas law. The gas is cooled and compressed to a volume of 50 ml, at 33 ☌. g of Ar occupy at 0 ☌, 760 mm Hg? (Hint: what's special about 0 ☌, 760.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
